Which Numerical Setup Can Be Used to Calculate the Atomic Mass of the Element Bromine
Chemistry worksheet
Topic: Isotopes Atomic Mass
Isotopes Atomic Mass
1
The table below gives the diminutive mass and the abundance of the two naturally occurring isotopes of chlorine.

Which numerical setup can be used to summate the atomic mass of the chemical element chlorine?
(1) (34.97 u)(75.76) + (36.97 u)(24.24)
(two) (34.97 u)(0.2424) + (36.97 u)(0.7576)
(iii) (34.97 u)(0.7576) + (36.97 u)(0.2424)
(iv) (34.97 u)(24.24) + (36.97 u)(75.76)
2
The tabular array below shows the atomic mass and natural affluence of the ii naturally occurring isotopes of lithium.
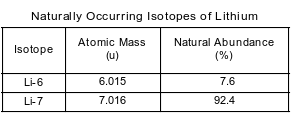
Which numerical setup tin can exist used to determine the atomic mass of naturally occurring lithium?
(i) (7.6)(6.015 u) + (92.4)(seven.016 u)
(2) (0.076)(half dozen.015 u) + (0.924)(7.016 u)
(3) ![]()
(4) ![]()
three
Naturally occurring gallium is a mixture of isotopes that contains 60.11% of Ga-69 (atomic mass = 68.93 u) and 39.89% of Ga-71 (atomic mass = 70.92 u). Which numerical setup can be used to determine the atomic mass of naturally occurring gallium?
(1) ![]()
(2) ![]()
(iii) (68.93 u)(0.6011) + (70.92 u)(0.3989)
(4) (68.93 u)(39.89) + (lxx.92 u)(sixty.eleven)
four
The table below gives the atomic mass and the abundance of the two naturally occurring isotopes of boron.
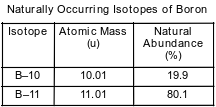
Which numerical setup can be used to deter- mine the atomic mass of the element boron?
(1) ![]()
(2) ![]()
(3) ![]()
(4) ![]()
v
The numbers of protons and neutrons in each of four different atoms are shown in the tabular array below.
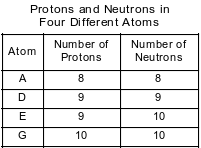
Which two atoms stand for isotopes of the same element?
(1) A and D
(2) A and Thousand
(3) E and D
(4) E and Thou
6
Which notations stand for atoms that accept the same number of protons only a different number of neutrons?
(1) H-3 and He-3
(2) Southward-32 and S-32
(3) Cl-35 and Cl-37
(4) Ga-70 and Ge-73
seven
The weighted average of the atomic masses of the naturally occuring isotopes of an element is the
(1) atomic mass of the element
(2) atomic number of the element
(iii) mass number of each isotope
(4) formula mass of each isotope
viii
The tabular array below gives the diminutive mass and the abundance of the two naturally occurring isotopes of bromine.

Which numerical setup can exist used to calculate the atomic mass of the element bromine?
(1) (78.92 u)(l.69) + (80.92 u)(49.31)
(ii) (78.92 u)(49.31) + (80.92 u)(l.69)
(3) (78.92 u)(0.5069) + (80.92 u)(0.4931)
(4) (78.92 u)(0.4931) + (80.92 u)(0.5069)
Base your answers to questions nine on the data below and on your noesis of chemical science.
Some isotopes of neon are Ne-19, Ne-20, Ne-21, Ne-22, and Ne-24. The neon-24 decays by beta emission. The diminutive mass and natural abundance for the naturally occurring isotopes of neon are shown in the table beneath.
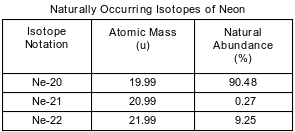
nine
Land the number of neutrons in an atom of Ne-20 and the number of neutrons in an atom of Ne-22.
Allow 1 credit. Acceptable responses include, but are not express to:
• Ne-20: 10
• Ne-22: 12
Base of operations your answers to questions x on the information below and on your noesis of chemistry.
The diagram below shows the kickoff 3 steps in the uranium-238 radioactive disuse series.
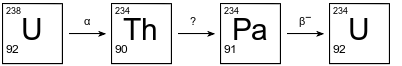
The decay mode for the first and 3rd steps is shown above the arrows. The decay mode for the 2nd step is not shown in the diagram. Thorium-234 has a one-half-life of 24.10 days.
10
Explain, in terms of neutrons and protons, why U-238 and U-234 are different isotopes of uranium.
Permit 1 credit. Acceptable responses include, but are not limited to:
• An atom of U-238 has 92 protons and 146 neutrons. An cantlet of U-234 besides has 92 protons but has 142 neutrons.
• These two atoms have the aforementioned number of protons just a different number of neutrons.
Base your answers to questions 11 on the data below and on your knowledge of chemistry.
The table below shows information for 3 isotopes of the same element.
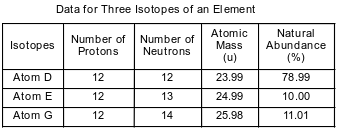
11
Explain, in terms of subatomic particles, why these three isotopes stand for the same element.
Allow i credit. Acceptable responses include, but are not limited to:
• Atoms of isotopes D, E and K have the same number of protons.
• They each have 12 protons.
Base your answers to questions 12 on the information below and on your noesis of chemistry.
The only naturally occurring isotopes of nitrogen are N-14 and North-xv.
12
Based on the atomic mass of the element nitrogen on the Periodic Table, compare the relative abundances of the naturally occurring isotopes of nitrogen.
Allow i credit. Acceptable responses include, only are not limited to:
• Nitrogen-xiv is more abundant than nitrogen-xv.
• There are fewer Due north-15 atoms.
Base your answers to questions 13 on the information below and on your noesis of chemistry.
Iodine has many isotopes, merely only iodine-127 is stable and is found in nature. One radioactive iodine isotope, I-108, decays by blastoff particle emission. Iodine-131 is also radioactive and has many of import medical uses.
13
Explicate, in terms of protons and neutrons, why I-127 and I-131 are different isotopes of iodine.
Let 1 credit. Acceptable responses include, but are not limited to:
• I-127 atoms and I-131 atoms have the same number of protons, but different numbers of neutrons.
• Both have 53 p, but I-127 has 74 n while I-131 has 78 n.
• They have the same atomic number just different mass numbers.
• same atomic number but unlike numbers of neutrons
• The only difference is the number of neutrons.
Base your answers to questions 14 on the information below and on your knowledge of chemistry.
Cobalt-60 is an artifi cial isotope of Co-59. The incomplete equation for the decay of cobalt-60, including beta and gamma emissions, is shown below.
![]()
14
Explicate, in terms of both protons and neutrons, why Co-59 and Co-threescore are isotopes of cobalt.
Permit ane credit. Acceptable responses include, just are not limited to:
• Both atoms take 27 protons, but Co-59 atoms each have 32 neutrons and Co-60 atoms each take 33 neutrons.
• aforementioned number of protons, different number of neutrons
Base your answers to questions 15 on the information below.
The element boron, a trace chemical element in Earth's crust, is found in foods produced from plants. Boron has just two naturally occurring stable isotopes, boron-10 and boron-xi.
15
Compare the abundance of the two naturally occurring isotopes of boron.
Allow i credit. Acceptable responses include, but are not limited to:
• Boron-11 is nigh four times more arable than boron-10.
• The B-10 is less abundant.
Source: https://www.edusofttech.com/chemistry/worksheet-quiz/isotopes-atomic-mass/
0 Response to "Which Numerical Setup Can Be Used to Calculate the Atomic Mass of the Element Bromine"
Post a Comment